Salmonella Antisera
SSI Diagnostica produces more than 200 different Salmonella antisera, making it possible to type 99% of all Salmonella serotypes.

Salmonella Antisera
SSI Diagnostica has more than 75 years of experience with research and production of Salmonella antisera, and we offer a range of products with strong portfolio breadth and depth to facilitate accurate identification as specified by the Kaufmann-White Scheme.
Salmonella is often linked with food safety issues, and it is the leading cause of food poisoning. In compromised patients it can even lead to death. Therefore, it is important to get fast and reliable results.
View our videos on how to perform Slide agglutination and Phase Inversion below.
Slide Agglutination:
Phase Inversion:
Gold Standard in Serotyping
The Kauffmann–White Le Minor scheme describes the antigen composition of >2,500 identified Salmonella enterica serotypes. SSI Diagnostica produces more than 200 different Salmonella antisera, making it possible to type 99% of all Salmonella serotypes.
All our antisera are produced in accordance with “Guidelines for the preparation of Salmonella antisera” (ref.: WHO Collaborating Centre for Reference and Research on Salmonella, Institute Pasteur, France).
Quality and Storage
SSI Diagnostica ready-to-use antisera are high quality, CE-marked and produced in accordance with DS/EN ISO 13485. The shelf life is 4 years from the date of production, and a minimum of 2 years from the delivery date. Sodium azide 0.09% is added to all Salmonella antisera as a preservative.
The antisera must be stored at 2–8°C
Salmonella Antisera Solutions
Salmonella O and Vi antisera
Poly and Vi Salmonella antisera for serological confirmation by slide agglutination.
Read more
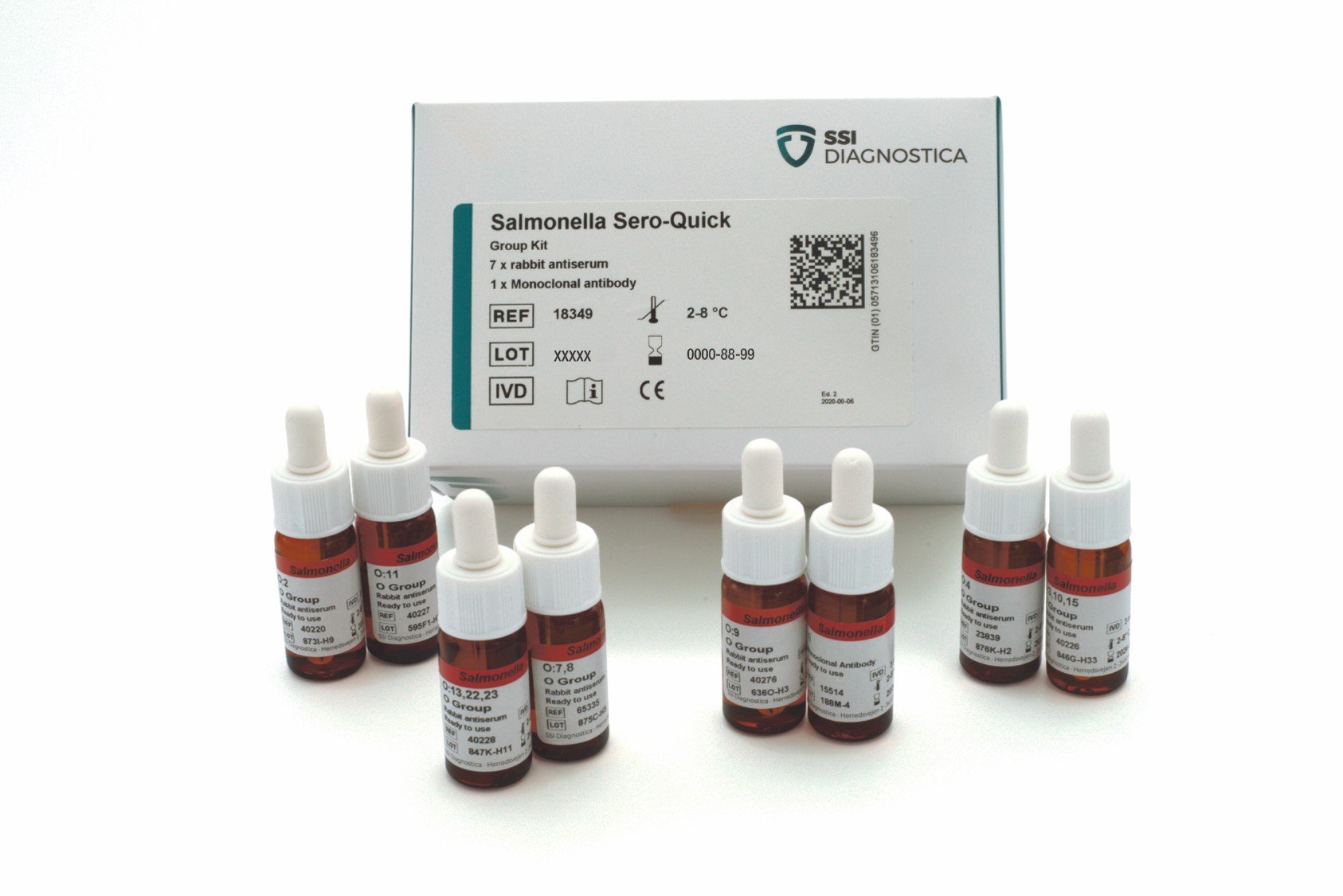
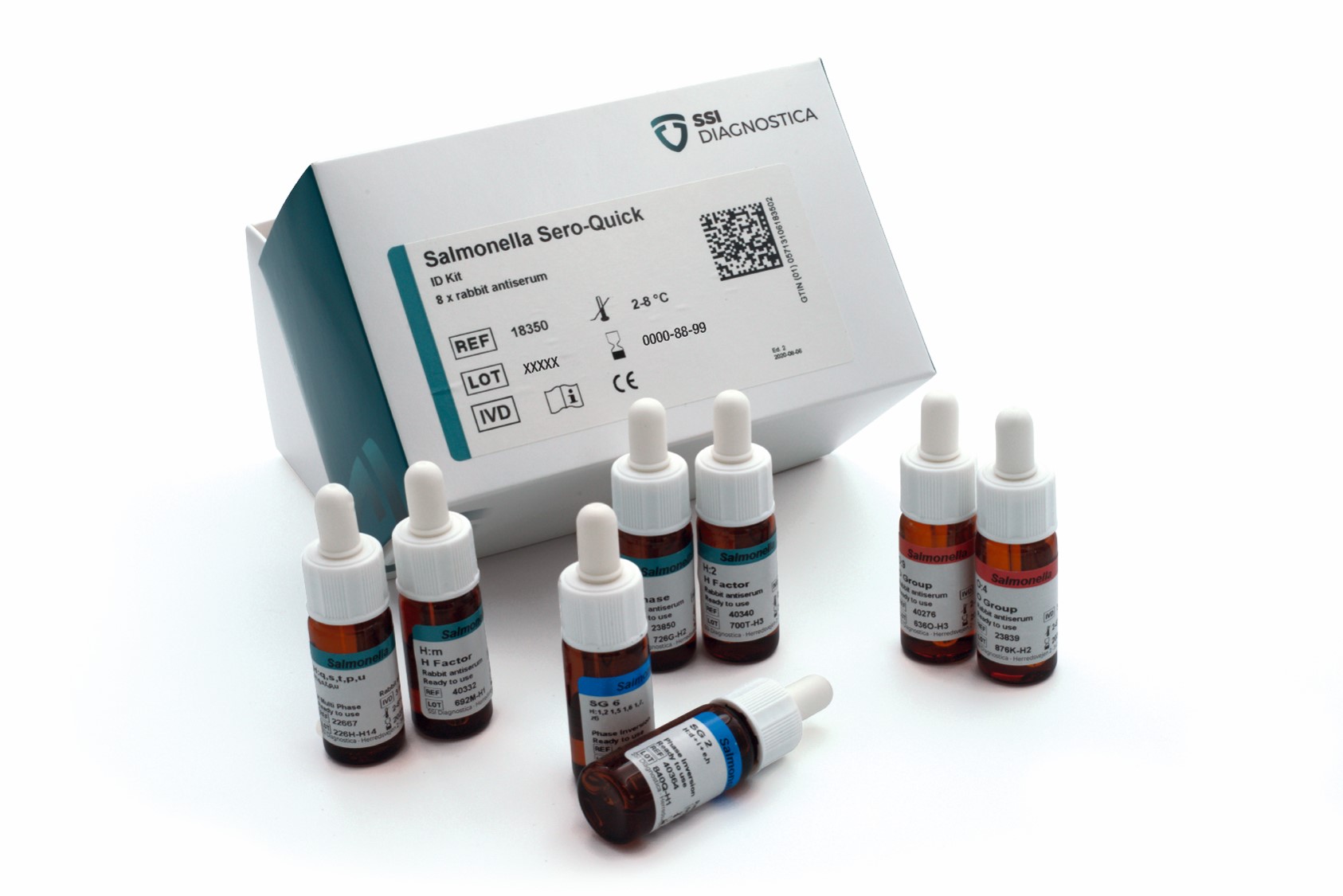
Salmonella Sero-Quick Group Kit
The Salmonella Sero-Quick Group Kit identifies Salmonella isolates to serogroup level.
Read more
Product range
SSI Diagnostica Salmonella antisera can identify O and H antigens and the capsular antigen Vi. Our Salmonella antisera are grouped as follows:
| Sera | Use |
|---|---|
| O poly antisera | Verification of genus (Salmonella enterica) |
| O group pool antisera | For multi-group screening |
| O group antisera | To determine O group |
| O factor antisera | To determine O factor |
| H poly antisera | Verification of genus (Salmonella enterica) |
| H phase pool antisera | For multiphase screening |
| H phase antisera | To determine H phase |
| H factor antisera | To determine H factor |
| H:R-phase antisera | To determine R phases |
| H phase inversions antisera | For phase inversion in swarm agar plates |
| Vi antiserum | To investigate the presence of Vi capsular antigen |
| Sero-Quick Group Kit | For quick serogrouping of Salmonella |
| Sero-Quick ID Kit | For quick identification of S. Enteritidis and S. Typhimurium |
Simpler for the better
If you work within the field of microbiology, you might be familiar with Occam’s Razor. At SSI Diagnostica, we champion a slightly adapted version of this principle:
The simplest solution is usually the best one.
Contact
Please, reach out if you have questions, comments or requests.